Eli Lilly & Co. v. Teva Parenteral Medicines, Inc. (Fed. Cir. 2012) | McDonnell Boehnen Hulbert & Berghoff LLP - JDSupra

Teva detiene producción en una de sus plantas farmacéuticas de Estados Unidos después de inspección de la FDA - enFarma
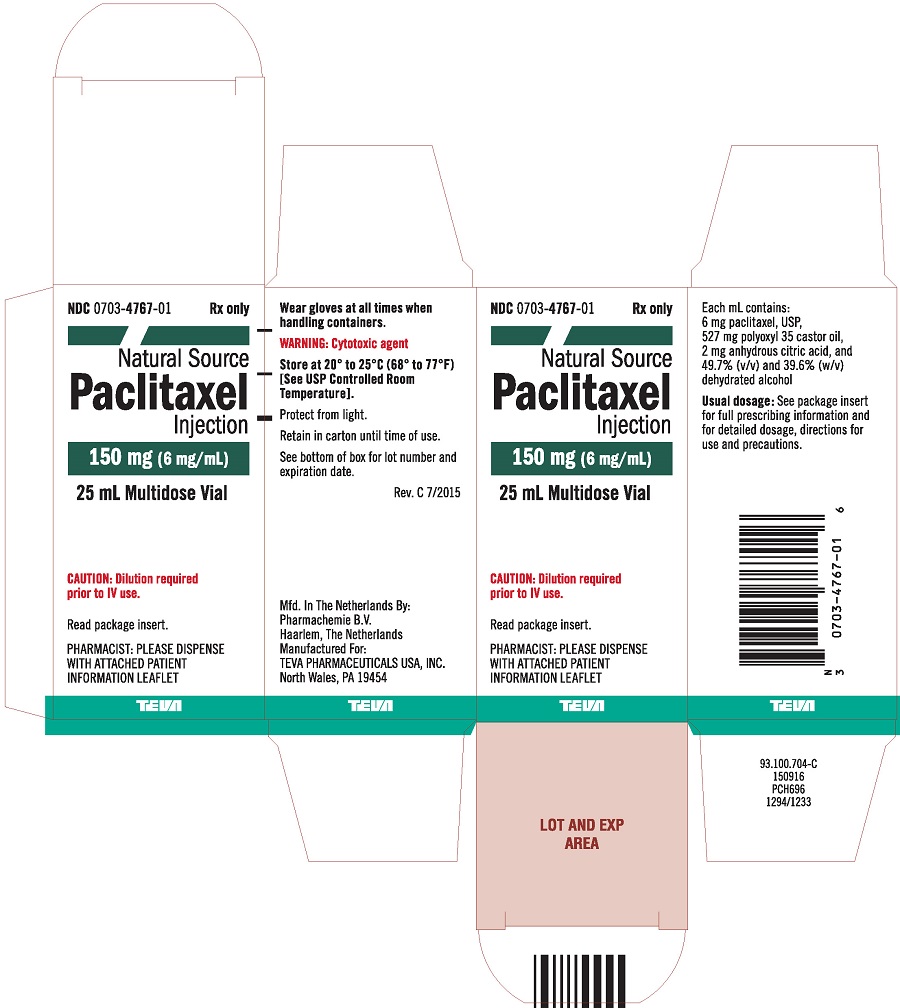
ANDA 77-453 Teva Parenteral Medicines,Inc. Attention: Susan O'Brien Director, Regulatory Affairs 19 Hughes Irvine, CA 92618-1
Page 1 of 5 Warning Letters > Teva Parenterals Medicines, Inc. 12/11/09 21.10.2010 http://www.fda.gov/ICECI/EnforcementAction
ZANOSAR- streptozocin powder, for solution Teva Parenteral Medicines, Inc. ---------- ZANOSAR (Streptozocin Sterile Powder) Pac

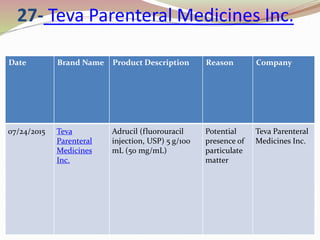
Teva Parenteral Medicines Initiates Voluntary Nationwide Recall of Select Lots of Adrucil® (fluorouracil injection, USP) 5 g/100 mL (50 mg/mL) Due to Particulate Matter | Business Wire

Teva Parenteral Medicines Initiates Voluntary Nationwide Recall of Select Lots of Adrucil® (fluorouracil injection, USP) 5 g/100 mL (50 mg/mL) Due to Particulate Matter | Business Wire







/cloudfront-us-east-2.images.arcpublishing.com/reuters/CCBS56SBRRKUDM7JEVVE3UOQEE.jpg)