Bacterial RTX Toxins Allow Acute ATP Release from Human Erythrocytes Directly through the Toxin Pore* - Journal of Biological Chemistry
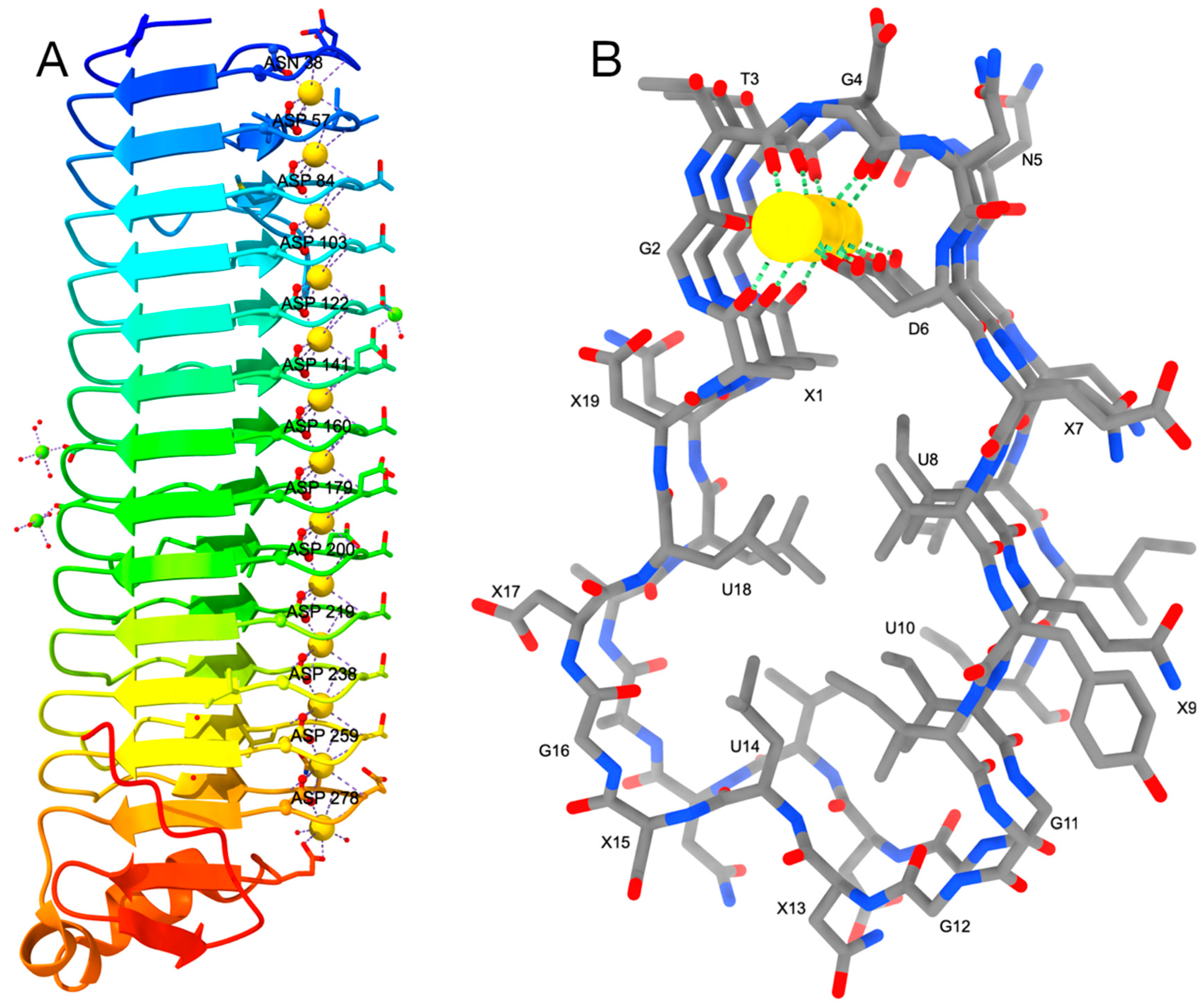
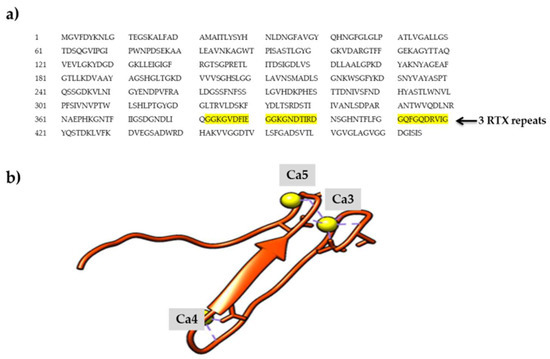
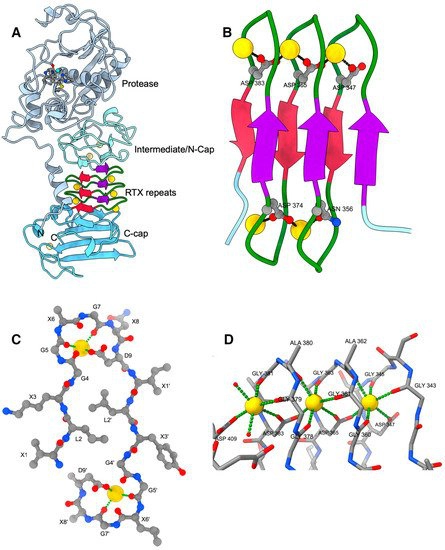
Toxins | Free Full-Text | Structure–Function Relationships of the Repeat Domains of RTX Toxins | HTML
Structural basis for antibody binding to adenylate cyclase toxin reveals RTX linkers as neutralization-sensitive epitopes | PLOS Pathogens
Membrane localization of the Repeats-in-Toxin (RTX) Leukotoxin (LtxA) produced by Aggregatibacter actinomycetemcomitans | PLOS ONE
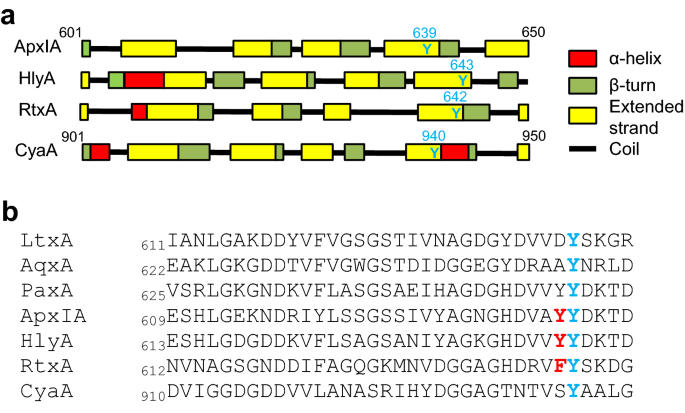
Different roles of conserved tyrosine residues of the acylated domains in folding and activity of RTX toxins | Scientific Reports

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect

Coordinated delivery and function of bacterial MARTX toxin effectors - Woida - 2018 - Molecular Microbiology - Wiley Online Library

Identification of a Vibrio cholerae RTX toxin gene cluster that is tightly linked to the cholera toxin prophage | PNAS

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect

In vivo covalent cross‐linking of cellular actin by the Vibrio cholerae RTX toxin | The EMBO Journal

The Vibrio cholerae MARTX toxin simultaneously induces actin collapse while silencing the inflammatory response to cytoskeletal damage | bioRxiv

Templated folding of the RTX domain of the bacterial toxin adenylate cyclase revealed by single molecule force spectroscopy | Nature Communications
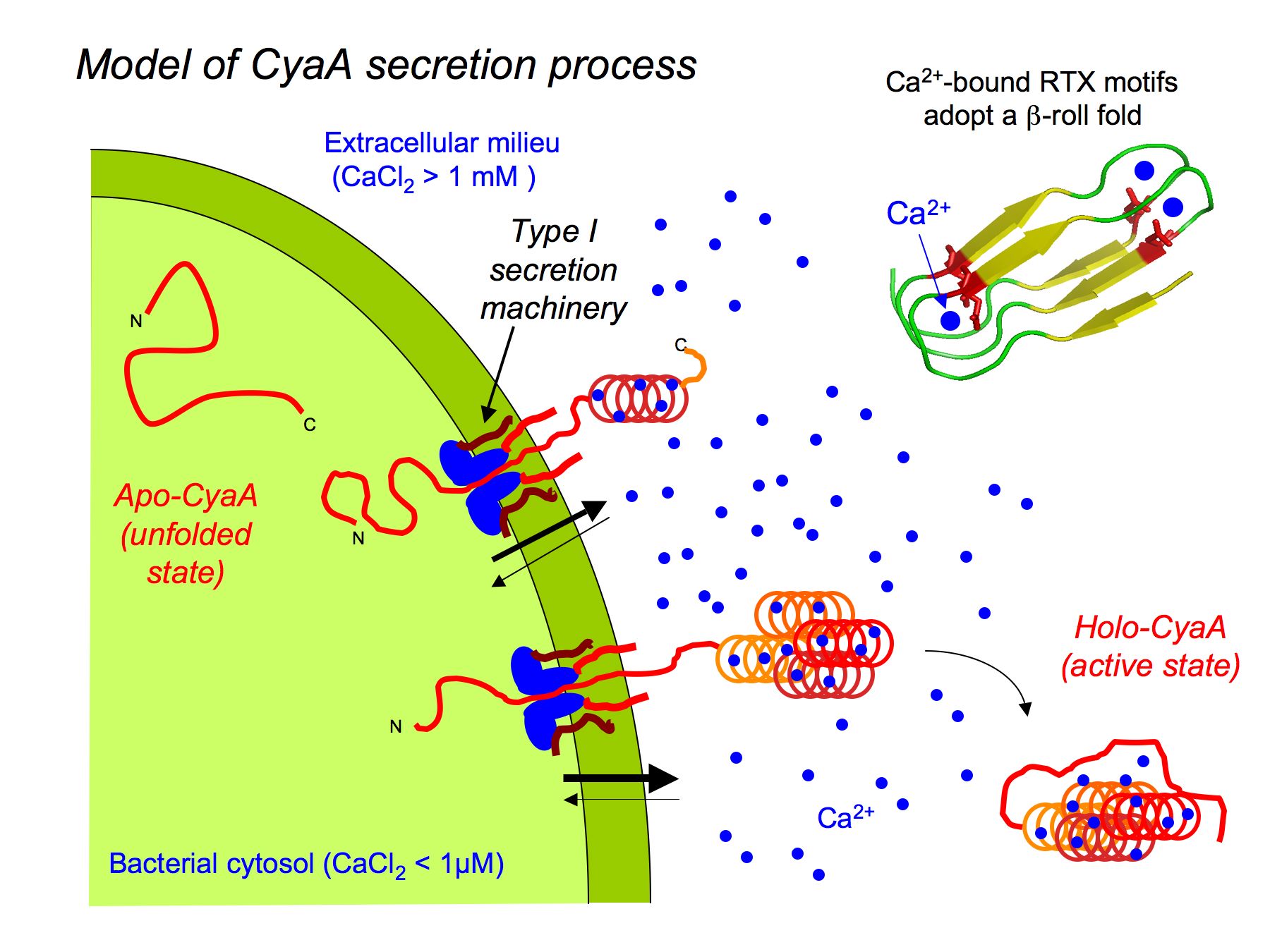
![PDF] Disorder-to-Order Transition in the CyaA Toxin RTX Domain: Implications for Toxin Secretion | Semantic Scholar PDF] Disorder-to-Order Transition in the CyaA Toxin RTX Domain: Implications for Toxin Secretion | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a2a64dd1c479adcb98ca01116219b7682c36185f/3-Figure1-1.png)